|
1/2/2023 0 Comments Iso 13485 requirements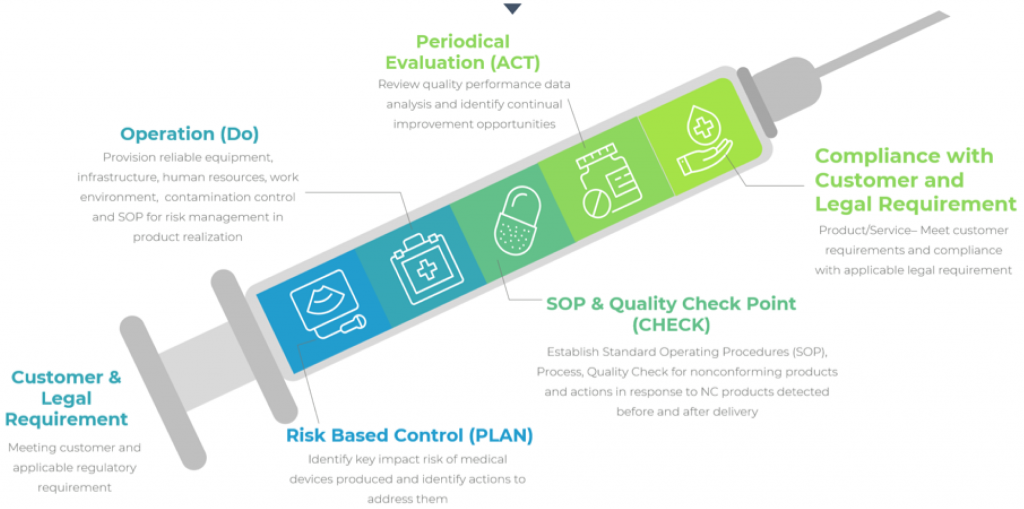
While not mandatory, an organisation may find difficulty operating in some markets or getting their product recognised without it. ISO 13485 and document control for programsĬustomer Property - ISO 13485:2016 Clause 7.5.In a nutshell, no, ISO 13485 is not mandatory, however, certification is extremely important as it harmonises your practices with legal and regulatory requirements that exist across the country, the EU and the rest of the globe. Question on using audit checklist ISO 13485:2016 ISO 13485-paragraphs for a SaaS SAMD needed or not? Question regarding where "validations" fit according to ISO 13485:2016Īny template/ form of Monitoring and Measurement of Processes and product to ISO 13485? How to calculate Effective Number of People for ISO 13485 Certification? Medical Device and FDA Regulations and Standards News ISO 13485:2015 Software Validation IQ/OQ/PQ Notified Bodies - ISO 13485 & MDR Technical Filesĭeviations - Where in ISO 13485 deviations are covered? worth it?Įlectronic Signatures - Non-Conformance - ISO 13485:2016 ISO 13485 certification prior to Medical Device Manufacturing. ISO 13485- 8.3.1 Non-conforming material high volumeĬontract Manufacturer as Design Owner ISO 13485 ISO 13485 Certification - Class II product - Fluoride-free toothpaste - Records ISO 13485 sample document or template - Class II medical device ISO 9000, ISO 9001, and ISO 9004 Quality Management Systems StandardsĬAP for Medical Device Class Is product - Slightly confused - ISO 13485 ISO 13485 Risk Analysis - Class II Medical Device Engineering Changesĭistributors for class II medical devices in US - Do we need ISO 9001 or ISO 13485 Medical Device Class IIa Nonsterile ISO 13485 AlternativesĬlass IIb Medical Device using Annex II route - Need to Update ISO 13485 Scope? Training - Internal, External, Online and Distance Learning ISO 13485 Book and Training Class Recommendations wanted ISO 13485 Class I Medical Device Design Exclusion ISO 13485 Contract Manufacturer & Class II Devices ISO 13485 and Class 1 Medical Device - FDA thinkingĬlass I Technical File reviewed during ISO 13485 Certification Audit by NBĮxtent of Product Traceability Required by ISO 13485 (Low Level Class I Devices) ISO 13485 requirements for Stand Alone Medical Device Software (Class II a) 
Pre-Work and Study for ISO 13485:2016 Lead Auditor Class ISO 13485:2016 Self Certification for Class I (annex 9) Stand-Alone Software? ISO 13485 certification advantages when I certify a Class 2a medical device ISO 13485 Required for CE Mark on Class I productĬE Marking (Conformité Européene) / CB Scheme Medical device manufacturing (class 5:2016) - Is a Deviation allowed? MDR ISO 13485 for Class I sw/hw company to be class II due dates ISO 14971 applied to ISO 13485? Low risk class 1 devices ISO 13485 Requirement in Australia/NZ for class 1? ISO 13485:2016 - Medical Device Quality Management SystemsĬontamination Control - Class Is medical devices (Clause 6.4.5:2016 (E)) While you are upgrading the cleanroom, keep your eyes open for the additional low hanging fruit. Show me a dingy looking cleanroom with an acceptable particle count, I'll consider having it measured myself or shopping elsewhere. Note lastly that appearance affects not only your employees, but your customer's impression as well. A minor point to be sure, but get the mileage where you can. Note also that dress is a multifaceted can affect employee attitude and expected behavior as well. Make sure to measure the particle count with people actively working in the room, otherwise you just have a meaningless number. Use the tools to achieve the particle count requirement. Gowning and dress is simply a tool, as are airflow, bioburden (love that term.I'm a Bioburden!.I always thought I was just plain human), # of inlets, location of returns, inlet & return balancing, local process particulate capture, etc. Check out the requirements for 0.1um, 0.2um also. Many customers only care about 0.5um and up.but some care about the whole spread. There are various requirements for different particle sizes. The requirement is based on partcle count.not how you achieve it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
